Translate this page into:
Illuminating the obscure: An imaging study of male breast diseases
*Corresponding author: Pratibha Issar, Department of Radiodiagnosis, Jawahar Lal Nehru Hospital and Research Centre, Chhattisgarh, India. pratibhaissar@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Issar P, Desai S, Ravindranath M, Sahu P. Illuminating the obscure: An imaging study of male breast diseases. Indian J Breast Imaging. 2024;2:44-52. doi: 10.25259/IJBI_5_2023
Abstract
Male breasts mainly have fatty tissue with few ducts and stroma, in contrast to women’s breasts, which predominantly have ducts, stroma, and glandular tissue. This requires knowledge of normal male breast anatomy and physiological and pathological changes in the male breast. This article deals with normal anatomy and a spectrum of diseases involving the male breast.
Keywords
Male breast
Mammography
Ultrasound
Magnetic resonance imaging
Introduction
The proportion of male patients presenting with breast complaints is relatively small compared to female patients. They comprise approximately 1% of new breast cancer cases. Awareness of various imaging findings in male diseases and their differential diagnosis is essential for timely, appropriate treatment. This article provides an overview of the normal male breast anatomy, imaging protocols, and various conditions involving the male breast.[1]
Male Breast Anatomy
The male breasts extend from the second rib to sixth rib craniocaudally, and from the sternum to midaxillary line mediolaterally like the female breast.
At birth, both sexes have identical mammary glands. During the peripubertal period, estrogen stimulates ductal proliferation, branching, and growth. Progesterone causes stromal development and maturation of Terminal Ductal Lobular Unit (TDLU). A significant increase in testosterone levels in the male breast causes ductal involution and atrophy.
Compared to the female breast, the male breast lacks the Cooper ligament and has an involuted ductal system with rare TDLU development, smaller stroma, and more prominent pectorals muscles. Ductal malignancies are rarely seen and are located near the nipple-areolar complex. Routine screening for breast cancer in males is not deemed necessary due to the low occurrence rate of this disease in the male population. Due to delay in diagnosis, most of them present with advanced-stage axillary lymphadenopathy in nearly 50% of cases. Microcalcification, which shows ductal involvement, is rare in male breast cancer. Involuted ductal structure and a rare development of TDLU causes fibroepithelial (Fibroadenoma, Phyllodes tumor), lobular (lobular carcinoma in situ, invasive lobular carcinoma) and fibrocystic changes to be uncommon in the male breast.[2]
Among all imaging modalities, mammography is the modality of choice in patients with suspicious findings on physical examination followed by ultrasound (US) as next step for inconclusive mammograms. ACR appropriateness criteria suggest the US as first-line imaging below 25 years old and mammography after 25 years old.[3]
In craniocaudal and mediolateral oblique views, a typical male breast has a modest nipple-areolar complex, homogeneously radiolucent adipose tissue, and a prominent radiopaque pectoralis muscle [Figure 1]. Due to the well developed pectoral muscles, breast tissue can get obscured on mammogram. In such scenarios, patients should be asked to contract the muscle and breast tissue should be pulled forward to obtain an appropriate image.
- Difference between female and male breasts, (a) a mediolateral oblique (MLO) mammogram of a typical female breast showing well-developed fibroglandular structures (white arrow). (b) MLO view of a normal male breast showing the rudimentary form of the male breast with involuted fibroglandular tissue (small white arrow) and prominent pectoral muscle (large white arrow).
Ultrasound can evaluate the nipple mass relationship and axillary nodal involvement. It also helps assess the lesion’s vascularity, and sono-elastography can be used as an auxiliary modality.[4]
Magnetic resonance imaging (MRI) of the male breast can be utilized to assess chest wall involvement, post-operative residual disease, and response to chemotherapy. A biopsy is required for pathological diagnosis. Due to the reduced size of male breasts, US-guided biopsy should be preferred over stereotactic biopsies.[5]
Gynecomastia
Gynecomastia is caused by the growth of ductal and stromal components and is often characterized by unilateral or bilateral breast enlargement and localized discomfort. Etiology includes physiological, endocrine, metabolic, paraneoplastic, liver and kidney disease-related, idiopathic, and drug-induced factors. It can be seen in neonates, adolescents during puberty, and older men with increased estrogen and decreased testosterone levels. Nodular, dendritic, and diffuse glandular forms are the three characteristic patterns of gynecomastia.[6]
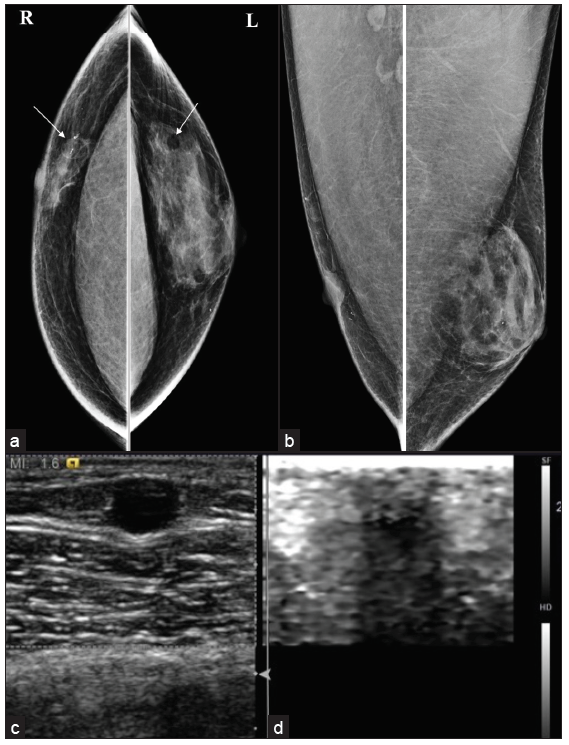
In the nodular form, ductal hyperplasia and cellular proliferative stroma are prominent. It corresponds to a duration of less than one year and is reversible if the underlying cause is eliminated. This form is often seen as fan-shape or nodular opacity in retroareolar location on mammography. US may illustrate it as a hypervascular and hypoechoic tissue which is disk shaped in subareolar location.
The dendritic form results from periductal stromal fibrosis and minimal ductal hyperplasia of more than one year with irreversible changes. On mammography, it is seen as flame-shaped subareolar opacity, that may have extension toward upper-outer quadrants and subareolar serpiginous tissue with star-shaped borders and spider-like appearance in the US [Figure 2]. The diffuse form is related to high estrogen therapy or exogenous hormone usage. Transgender male breasts usually show this pattern and appear as heterogeneous enlarged breast tissue appearance in mammography. In the US, increased breast volume and echogenicity are seen without Cooper ligaments. Gynecomastia can recur after surgery if the excision is incomplete, underlying causes are not treated, or post-surgical use of anabolic steroids [Figure 3].
- MLO views of different types of gynecomastia (a) Nodular, (b) Dendritic & (c) Diffuse glandular. (d) Dendritic type of gynecomastia on ultrasound showing typical spider leg appearance with no color flow. (e) MRI showing no contrast enhancement. (f) Histopathology shows a prominent duct surrounded by myxomatous stroma suggestive of gynecomastia.
- Postoperative case of bilateral gynecomastia with (a, b) recurrence of gynecomastia in the left breast and bilateral oil cyst (white arrows) seen in mammo and (c, d) Ultrasound with elastography.
The most common differential diagnosis of gynecomastia is pseudo-gynecomastia and malignancy. Pseudo gynecomastia is diffuse adipose tissue proliferation without fibro glandular development and can be unilateral or bilateral [Figure 4]. It is usually seen in obese individuals or with a Neurofibromatosis type I history without a discrete mass on physical examination. Liposuction is the treatment for pseudo-gynecomastia, whereas surgical excision of glandular tissue is required for gynecomastia. In gynecomastia, central involvement usually occurs and may extend to the upper-outer quadrant. In contrast, malignancy may be eccentric and cause secondary symptoms like thickening of the skin, ulceration, retraction of nipple, increase in breast trabeculation, and axillary lymphadenopathy. Gynecomastia may be associated with malignancy but is not the causative factor.[7]
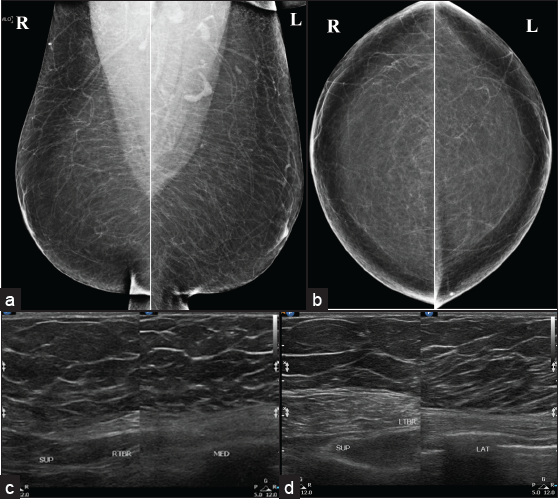
- Bilateral pesudo gynecomastia seen as (a, b) diffuse adipose tissue proliferation without fibroglandular component in mammo and (c, d) ultrasound.
Benign Neoplastic Breast Lesions
Lipoma
Lipoma is usually asymptomatic but may present as a non-tender lump. They present as well-defined, modestly encapsulated, radiolucent lesions on mammography [Figure 5] and avascular, oval-shaped, parallelly oriented, hyperechoic or iso-hypoechoic mass with thin echogenic capsules on the US.[8,9]
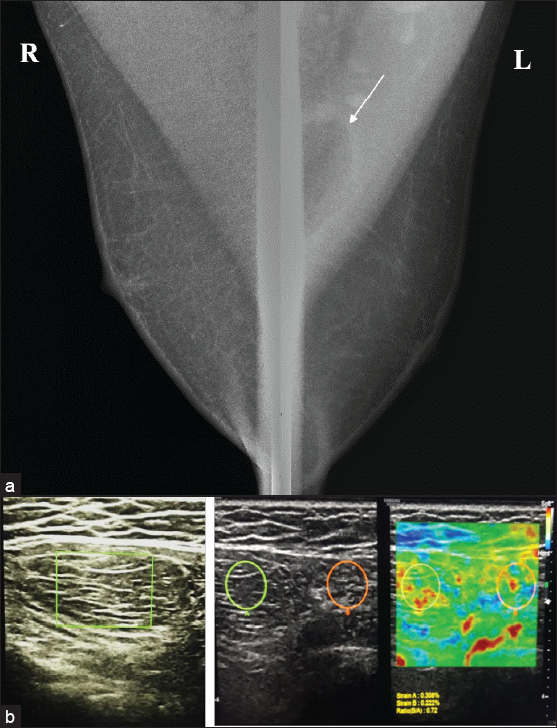
- (a) Mammogram showing an oval shape radiolucent mass (white arrow) within the left pectoral muscle. (b) On ultrasound, it is seen as an isoechoic lesion with multiple thin echogenic septations parallel to the skin surface. On elastography, it is seen as a soft tissue mass with an e-strain of .72, suggestive of intramuscular lipoma.
Myofibroblastoma
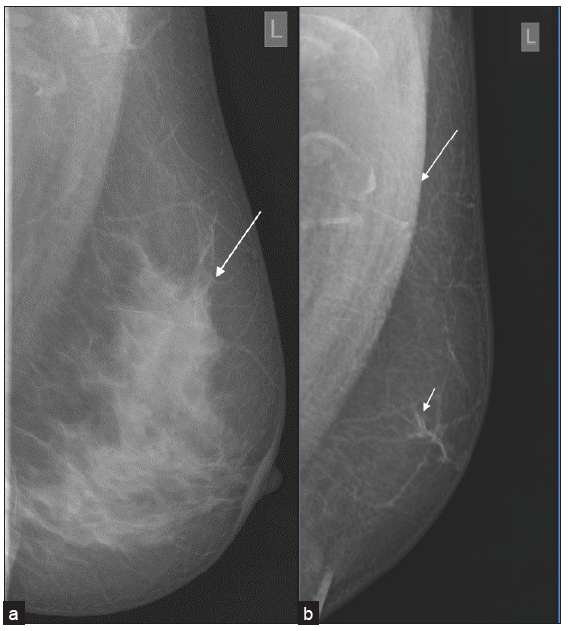
An uncommon benign stromal tumor containing spindle cells and hyalinized collagen, myofibroblastoma is seen as a mobile, well-defined, solid lump on physical examination. On mammography, they present as well-defined, encapsulated tumors without microcalcification [Figure 6]. In ultrasound mixed echo pattern can be seen.[10]
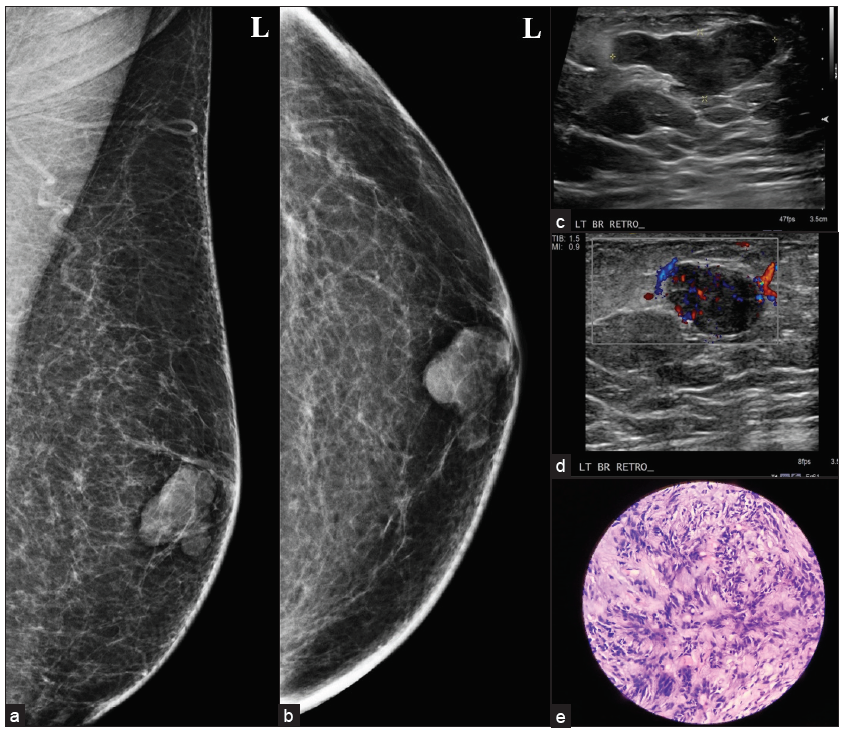
- A 50-year male with a history of a palpable lump in the left breast, (a, b) myofibroblastoma presenting as an oval circumscribed, equal density retro-areolar eccentric mass on MLO and CC view. (c, d) Ultrasound showed an oval, circumscribed, hypoechoic lesion with parallel orientation, no posterior features and areas of vascularity within it, (e) histopathology showing spindle cell lesion consistent with mammary type myofibroblastoma.
Fat Necrosis
This is a benign process. Usually, a history of trauma, surgery, or anticoagulation therapy is present. Mammography findings are related to the phase of the lesion. On Ultrasound, it may be seen as a avascular, circumscribed complex mass with or withoutfluid-fluid levels and internal septation. MRI, with the help of fat suppression images and contrast study [Figure 7], can also help detect these lesions.[11]
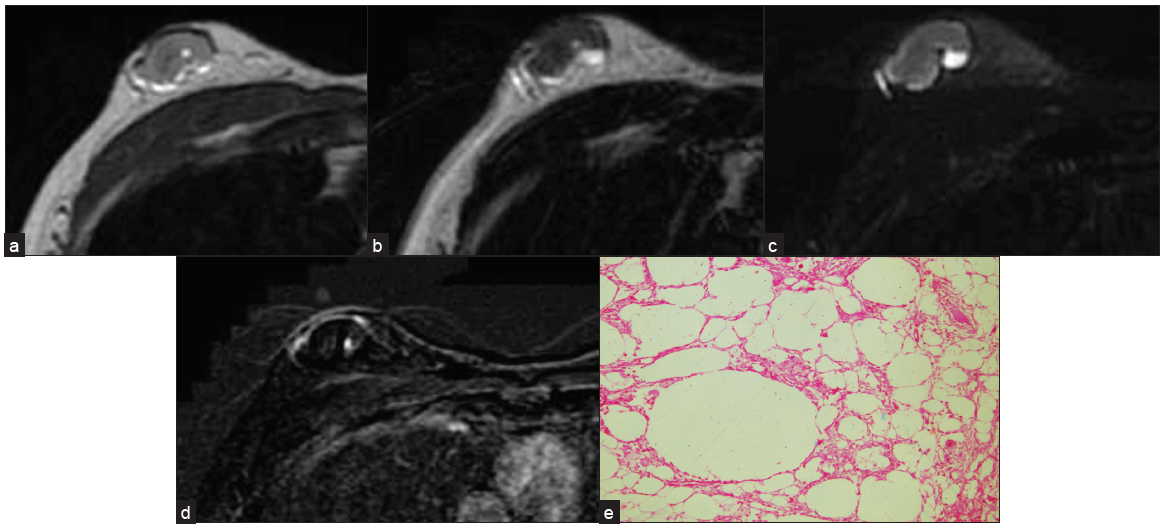
- A biopsy-proven case of fat necrosis seen as an oval circumscribed, heterogeneous mass on (a) T1 and (b) T2W1 (c) with surrounding hyperintense rim, which is suppressed on T2 Fat Sat image (d) and no contrast enhancement seen on subtraction image. (e) Anucleated adipocytes seen suggestive of fat necrosis.
Fibromatosis
Fibromatosis of the breast is a non-metastasizing benign but locally invasive stromal tumor which is characterized by the proliferation of fibroblastic and myofibroblastic cells. It is extremely rare in males and may mimic primary breast malignancy. Fibromatosis can be seen as high-density, non-calcified lesions with irregular shapes and spiculated margins on mammograms; which is not associated with adenopathy. MRI is useful for showing chest wall involvement, which is vital for surgical planning [Figure 8]. The diagnosis is confirmed by spindle cells and, on immunohistochemistry, the presence of smooth muscle Actin and Vimentin.[12]
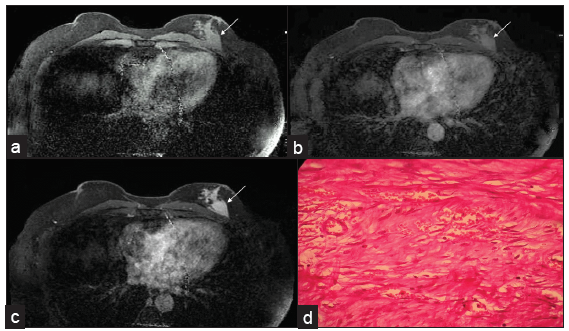
- MRI showing an irregular-shaped, spiculated margin mass (white arrows seen in a-c) in the left breast in the posterior third region adjacent to the pectoral muscle. (a) Pre-contrast image. Dynamic post contrast scans reveal progressive enhancement (b) early, (c) delayed images (d) Histology composed of bundles of long sweeping and intersecting spindle cells with collagen tissues suggestive of fibromatosis.
Malignant Lesions
Invasive Ductal Carcinoma (IDC)
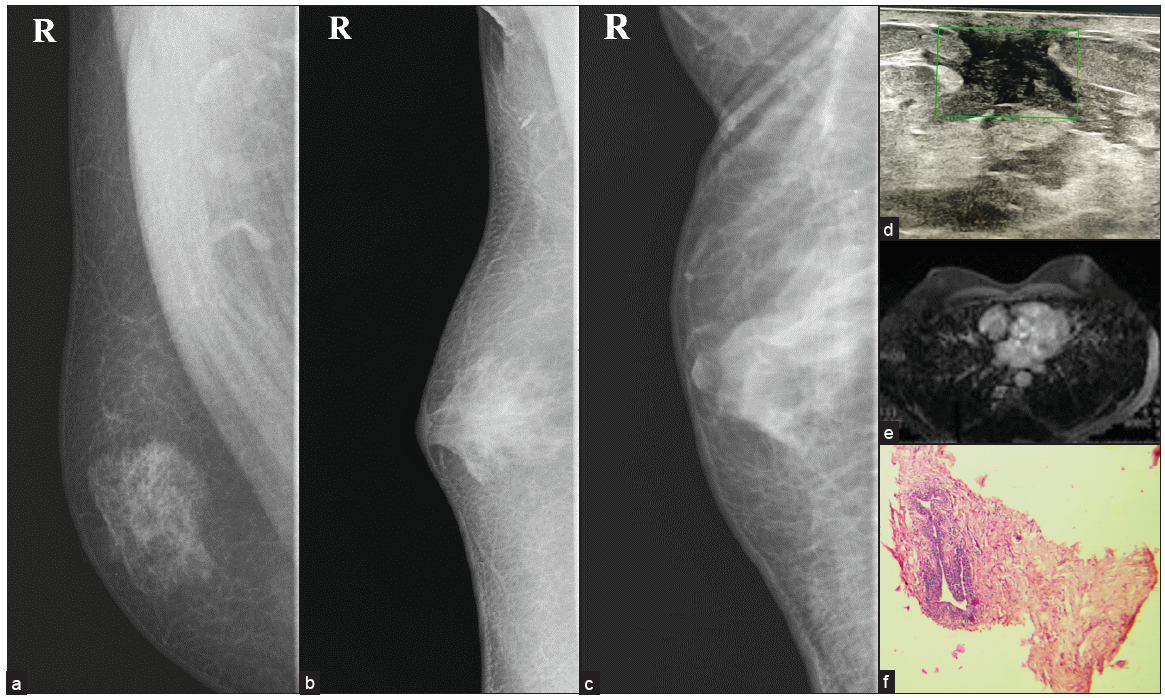
In male breasts, almost 80% of all cases belong to IDC “not otherwise specified type.” Multiple risk factors include family history, BRCA1 and BRCA2 mutations, Jewish, Egyptian, or West African descent, advanced age, exposure to ionizing radiation at a young age, cryptorchidism, and testicular injury. It usually presents as painless mass and can cause bloody nipple discharge.[13] The most common imaging finding is spiculated or irregular mass with or without associated pleomorphic calcification. Still, it can present with circumscribed masses that can be complex solid-cystic on ultrasound and have associated axillary lymphadenopathy [Figures 9 and 10].
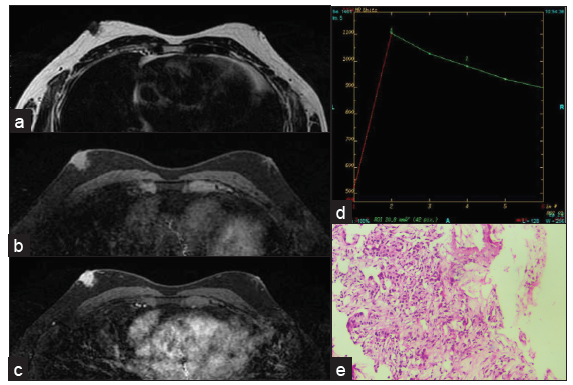
- Grade 1 invasive ductal carcinoma in a 54-year-old male presenting as a nodular mass in the subareolar region showing intense contrast enhancement with type III curve. (a) T2W1 (b) Pre-contrast (c) post-contrast (d) kinetic curve. (e) Histopathology showing grade 1 IDC.
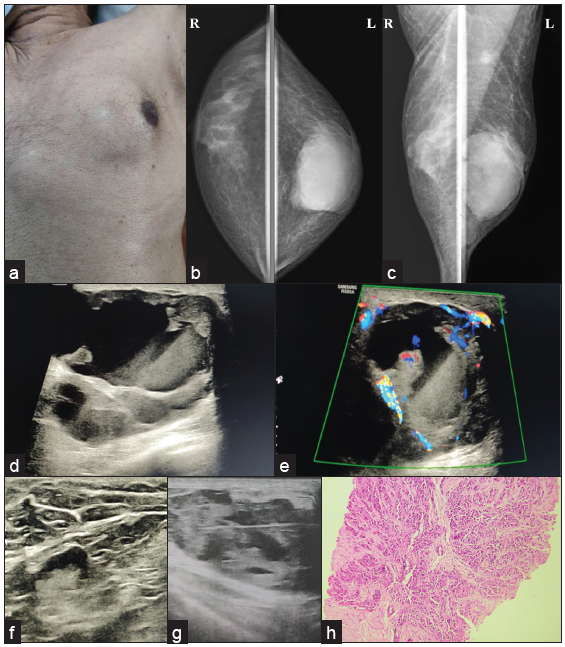
- A 72-year male with a complaint of a lump in left breast for six months, (a) displacing nipple-areolar complex toward left (b, c) MLO and CC view showing diffuse gynecomastia on the right side, and oval shape, circumscribed, high-density lesion involving left breast extending from subareolar to posterior third region with nipple retraction and overlying skin thickening along with axillary lymph node enlargement. (d, e) Ultrasound examination reveals a complex solid cystic lesion with peripheral and center vascularity (f) increased eccentric cortical thickness of lymph node, (g) and biopsy of the solid portion after fluid aspiration, turned out to be a case of (h) IDC grade III, seen in histopathology picture.
Papillary Carcinoma
Papillary Carcinoma exhibits a higher prevalence among males than females, with a ratio of 2:1. Mammography results often reveal well-defined, oval, lobulated, or irregular subareolar masses. Ill-defined borders point out the Infiltrative component.[14] On ultrasound, they appear as heterogeneous masses with solid cystic components [Figure 11].
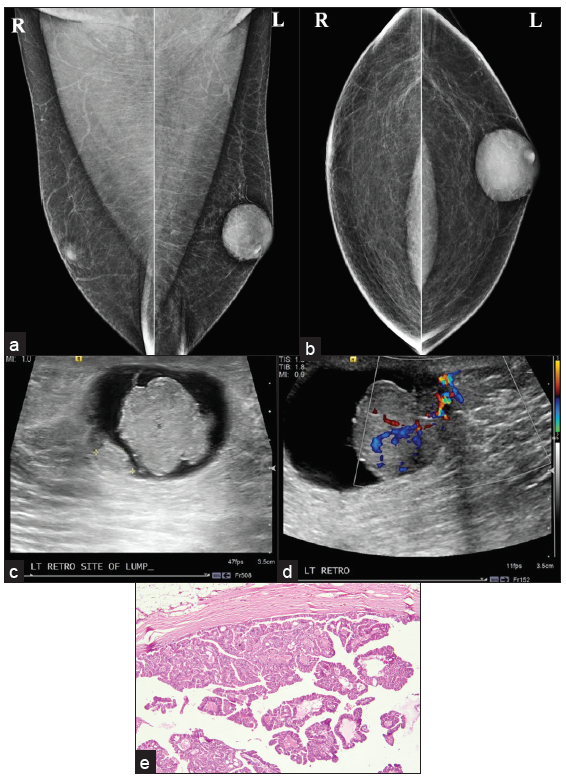
- Intracystic papillary carcinoma (a, b) MLO and CC view showing oval shape, circumscribed margin, high density subareolar eccentric lesions in the left breast. (c, d) Ultrasound shows irregular shape mass with indistinct margin, parallel in orientation, complex solid cystic pattern with areas of vascularity within the solid component, and posterior acoustic enhancement. (e) Histopathology showing intracystic tumor arranged in a tubulo-papillary pattern.
Plasmacytoma
Plasmacytoma of the breast can occur either as solitary extramedullary plasmacytoma (SEP) or as spectrum of dissemination of Multiple Myeloma. On ultrasound, they can appear as a mass with heterogeneous echotexture and few lobulations [Figure 12], so it is difficult to differentiate them from breast malignancy. Diagnosis is by Tru-Cut biopsy and plasmacytoid changes on histopathological examination and strong CD38 and CD138 expression.[15]
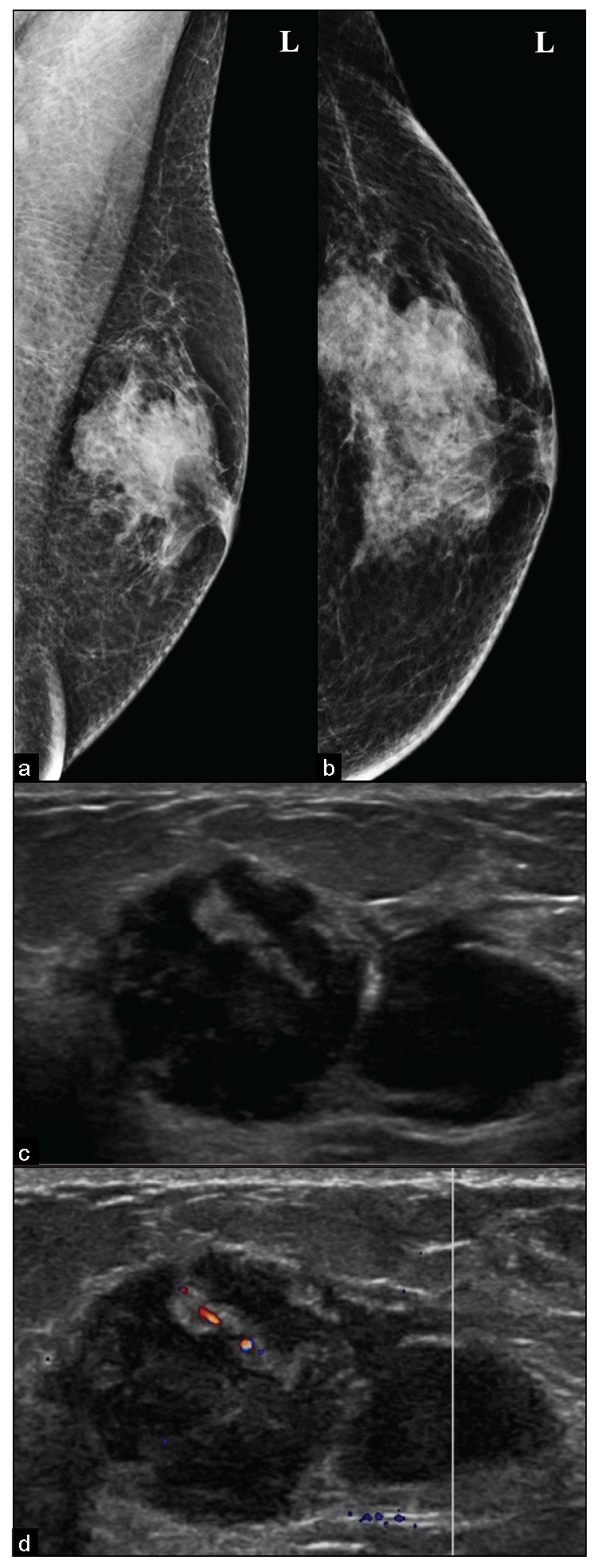
- Diagnosed case of Plasmacytoma, (a, b) presenting as an irregular microlobulated, high density, subareolar eccentric mass in the left breast, seen in mammography. (c, d) Ultrasound shows an irregular, microlobulated, heterogeneous mass with parallel in orientation and focal vascularity within it and no posterior features.
Synovial Sarcoma
Synovial Sarcoma is a malignant soft tissue tumor; few cases from the chest wall have been reported in the literature. Patients may present with chest pain, cough, dyspnea, reduced breath sounds, and weight loss.[16]
On computed tomography, it is seen as a heterogeneously enhancing mass with well-defined margins [Figure 13], cortical bone destruction, tumor calcification, and tumor infiltration of chest wall musculature, so they must be differentiated from breast malignancy.
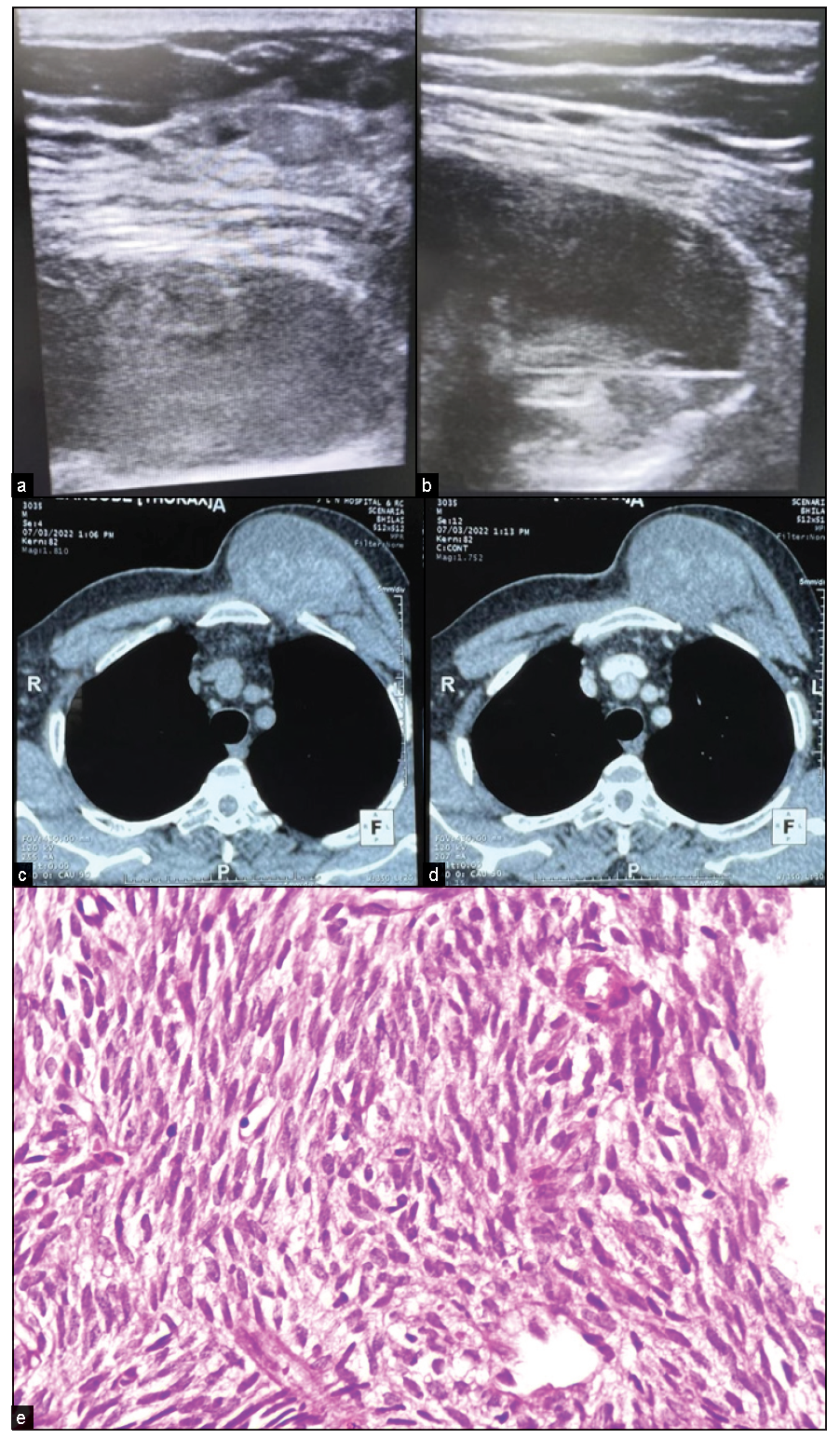
- A case of synovial sarcoma of the chest wall. (a, b) Ultrasound images show a mixed echogenic lesion posterior to the pectoral muscle with a biopsy needle in a solid portion. (c, d) CT image showing mildly enhancing heterogeneous lesion arising from chest wall pushing pectoralis major anterolaterally while maintaining fat planes. (e) Histopathology shows spindle cell tumors with skeletal muscle fragments. Tumor cells expressing Vimentin, CD56, CD99, and BCL2 suggestive of synovial sarcoma.
Axillary lymph node enlargement[17] can be seen in various conditions like reactive lymph node hyperplasia, atypical paracortical hyperplasia, Kikuchi disease, Castleman disease, tuberculosis, metastasis, and lymphoma [Figure 14].
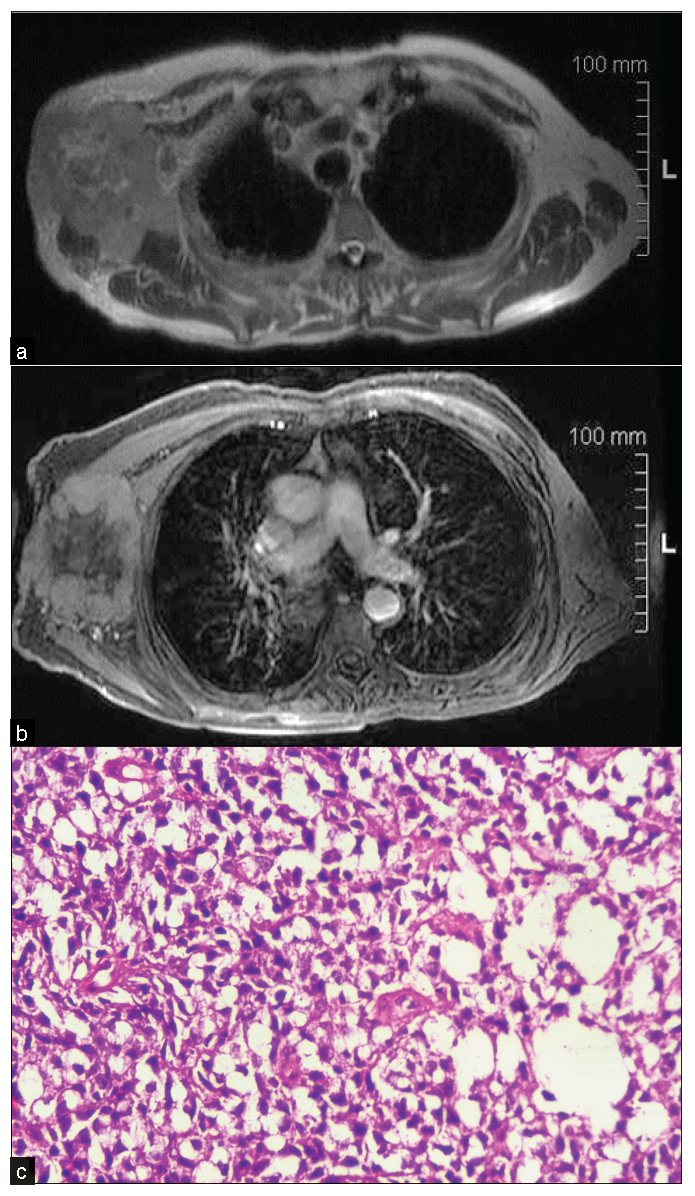
- An 80-year-old male with a complaint of the right axillary lump for two months. (a) T2 WI MRI showing irregular heterogeneous mass with irregular margin, (b) heterogeneous post-contrast enhancement in the right axilla, lateral to pectoral major and minor muscle with subcutaneous fat edema and involvement of overlying skin. (c) On biopsy, it was diagnosed as a case of non-Hodgkin’s B cell lymphoma with BCL2 and BCL6 positivity. Histopathology shows a nest of polygonal cells, having granular to clear cytoplasm and round to oval nucleus.
Conclusion
Understanding the anatomy and various benign and malignant male breast diseases can help us reach the correct diagnosis for proper treatment.
Ethical approval
The authors declare that they have taken the ethical approval and the approval number is IRB-2022/68-39.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Imaging findings and classification of the common and uncommon male breast diseases. Insights Imaging. 2020;11:27.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- ACR appropriateness criteria® evaluation of the symptomatic male breast. J Am Coll Radiol. 2018;15:S313-20.
- [CrossRef] [PubMed] [Google Scholar]
- Ultrasonography of the male breast. J Ultrasound. 2011;14:122-9.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Male breast magnetic resonance imaging: When is it helpful? Our experience over the last decade. Curr ProblDiagnRadiol. 2019;48:196-203.
- [CrossRef] [Google Scholar]
- Gynecomastia: Pathophysiology, evaluation, and management. Mayo Clin Proc. 2009;84:1010-5.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Male breast: Clinical and imaging evaluations of benign and malignant entities with histologic correlation. Am J Med. 2016;129:776-91.
- [CrossRef] [PubMed] [Google Scholar]
- Interpectoral lipoma mimicking a breast mass. J Breast Imaging. 2023;5:233-4.
- [CrossRef] [Google Scholar]
- Myofibroblastoma of the male breast: A rare entity with radiologic-pathologic correlation. Clin Imaging. 2017;42:109-12.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Fat Necrosis of the breast: A pictorial review of the mammographic, ultrasound, CT, and MRI findings with histopathologic correlation. Radiol Res Pract. 2015;2015:613139.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Breast fibromatosis: Radiologic–pathologic correlation. J Breast Imaging. 2021;3:597-602.
- [CrossRef] [PubMed] [Google Scholar]
- Male breast cancer with radiological and histopathological findings. SisliEtfalHastan Tip Bul. 2020;54:375-9.
- [CrossRef] [Google Scholar]
- Intracystic papillary carcinoma of the breast in males. Medicine (Baltimore). 2020;99:e20278.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Extramedullary plasmacytoma of the breast in a patient with Multiple Myeloma. Radiology Case. 2020;14:14-23.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Synovial sarcoma arising in the chest wall following ten years unexplained chest pain. OJRad2012. ;02:92-5.
- [CrossRef] [Google Scholar]
- Imaging-pathologic correlation of diseases in the axilla. Am J Roentgenol. 2013;200:W130-42.
- [CrossRef] [Google Scholar]







