Translate this page into:
A rare case of primary angiosarcoma of breast with histopathology correlation
*Corresponding author: Sanagapalli Lakshmi Naga Monica, Department of Radiology, NRI Medical College, Chinnakakani, Lakshmi Narayana Heights, Guntur, India. sln6monica@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Monica SLN, Rajani G, Inuganti RV. A rare case of primary angiosarcoma of breast with histopathological correlation. Indian J Breast Imaging. 2024;2:34-8. doi: 10.25259/IJBI_8_2023
Abstract
Primary angiosarcoma of the breast is a rare malignancy accounting for 0.05% of cases, seen in young females, and has atypical radiological presentation. We present a case of a 54-year-old female with complaints of a lump in the right breast for 5 months associated with pain, on and off fever. On ultrasound examination, the patient was found to have a BIRADS-IVb lesion. On further investigation using MRI, the lesion was found to have diffusion restriction with low ADC values, heterogenous enhancement, and type III enhancement curves, suggesting a malignant etiology. On histopathology, the lesion turned out to be angiosarcoma of intermediate grade and is positive for IHC CD 34.
Keywords
Angiosarcoma
Breast
Vascular lesions of the breast
Dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI).
Introduction
Angiosarcoma of the breast is one of the rare tumors and highly vascular tumors of the breast. It is of two types: primary and secondary. Primary angiosarcoma occurs in young women without any prior history or initiating event. Whereas secondary occurs in patients of prior radiotherapy or following treatment of breast carcinoma. Angiosarcoma of the breast has varied presentations, which can mislead the diagnosis to a benign lesion. Hence, it is worth considering MRI to differentiate the benign or malignant nature of the lesion and further aid in the early diagnosis and appropriate treatment.
Case Report
A 54-year-old female came to the surgery OPD with complaints of a lump in the right breast for 2 months. She also complains of pain, on and off fever. There were no similar complaints in the past or family history of breast carcinoma or any surgical history. No prior mammography was done. There was no obvious increase in the size of the mass. On clinical examination, there was a lump in the upper and outer quadrant of the right breast which was hard in consistency, tender and which was not freely mobile. There was focal skin thickening and no nipple retraction. Lymph nodes were not palpable.
The patient had severe pain for which she denied mammography. USG was advised. On USG, there was a well-defined, irregular, heterogeneous lesion with lobulated margins, oriented parallel to the breast parenchyma, showing posterior acoustic enhancement and significant internal vascularity. The lesion is about 4.5 cm away from the nipple. The lesion was seen extending up to the skin with focal skin thickening. Few cystic areas were seen within the lesion. There were no calcifications, architectural distortion, or nipple retraction. The lesion was highly suspicious for malignancy — BIRADS IVb lesion [Figure 1]. The left breast was normal. There was one lymph node with thickened cortex (cortex measuring 5 mm) found in the left axilla. Patient was further evaluated with DCE-MRI.
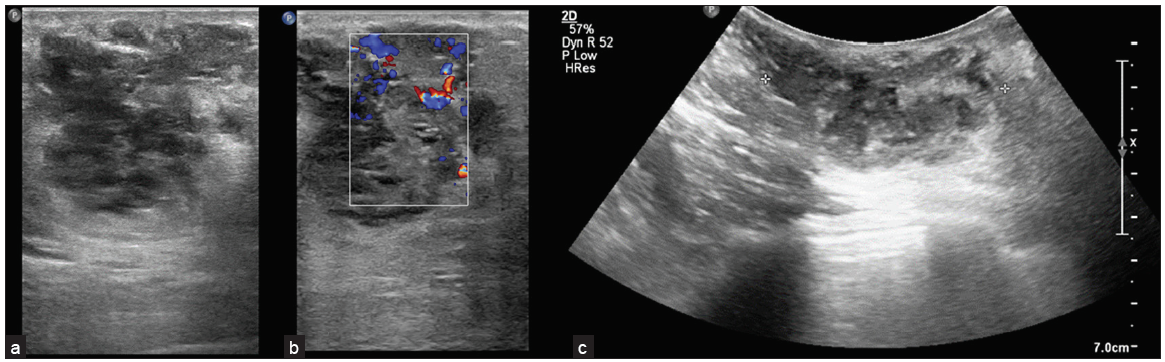
- (a) Irregular shaped lesion with microlobulated margins, oriented parallel to the breast parenchyma with few cystic areas within. (b) On colour doppler (white box: color box in the region of interest), there is significant internal vascularity. (c) The lesion shows posterior acoustic enhancement. Ill-defined, irregular, heterogeneously hypoechoic lesion with lobulated margins, showing posterior acoustic enhancement and significant internal vascularity is seen with few cystic areas in the lesion. The lesion was suspicious for malignancy --- Breast Imaging Reporting and Data system (BIRADS) IV lesion.
MRI was done using a 1.5T, GE machine. The protocol in our institute included Axial T1, T2, STIR images, Axial DWI, Sagittal STIR, and Dynamic contrast imaging (VIBRANT). 0.1 mmol/kg of gadobuterol is given with 20 mL of saline chase. Six phases of post-contrast T1 3D fat-suppressed images are taken for every 1 minute up to 6 minutes, after which images were analyzed with subtraction images and kinetic curves. MRI revealed a well-defined ovoid mass with lobulated and few irregular margins in the upper and outer quadrant, in posterior one-third depth, located approximately 5 cm from the nipple, and is seen extending up to skin surface in the lateral aspect associated with focal skin thickening. It is seen 4 mm away from the chest wall. The lesion is isointense on T1, intermediate on T2 and hyperintense on STIR with significant peritumoral edema, showing diffusion restriction with low ADC values of 0.2 × 10–6 mm/s [Figure 2]. On contrast administration, the lesion shows intense heterogeneous enhancement [Figure 3] and type III kinetic curves [Figure 4]. There is a single enlarged level I axillary lymph node with nodular cortical thickening (measuring 6 mm) and near total loss of fatty hilum in the left axilla. Findings were highly suspicious for malignancy – BIRADS IV. USG-guided biopsy was advised for further evaluation.
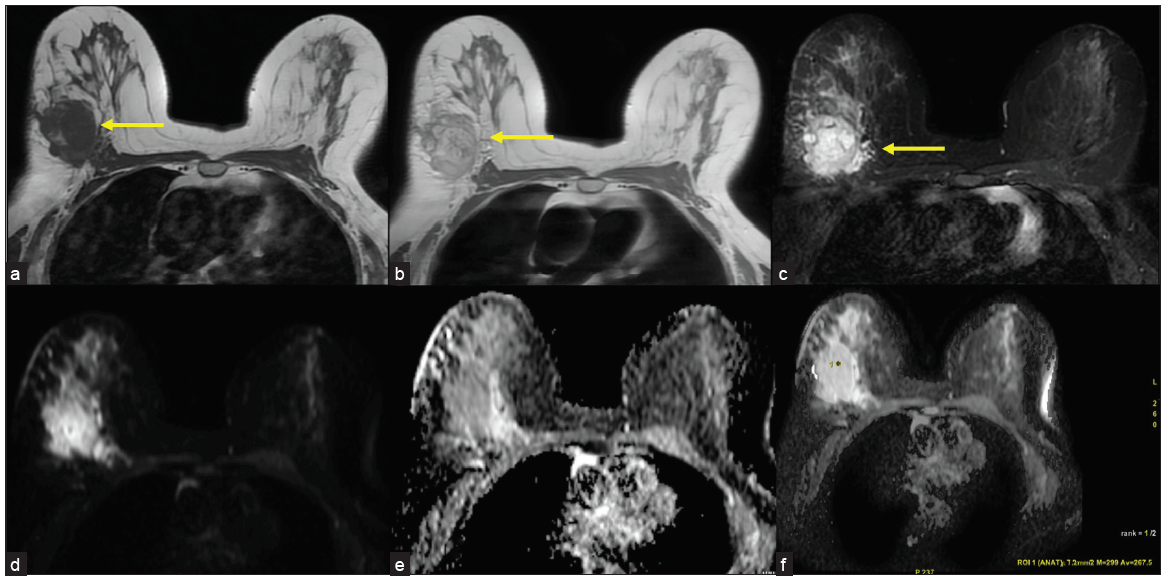
- (a) Axial T1 images showing hypointense lesion (yellow arrow) in the upper and outer quadrant of right breast. (b) Axial T2 images showing hyperintense lesion (yellow arrow). (c) Axial STIR images showing hyperintense lesion with significant edema (yellow arrow). (d-f) Lesion showing diffusion restriction with low ADC values of 0.2 × 10-6 mm/s.
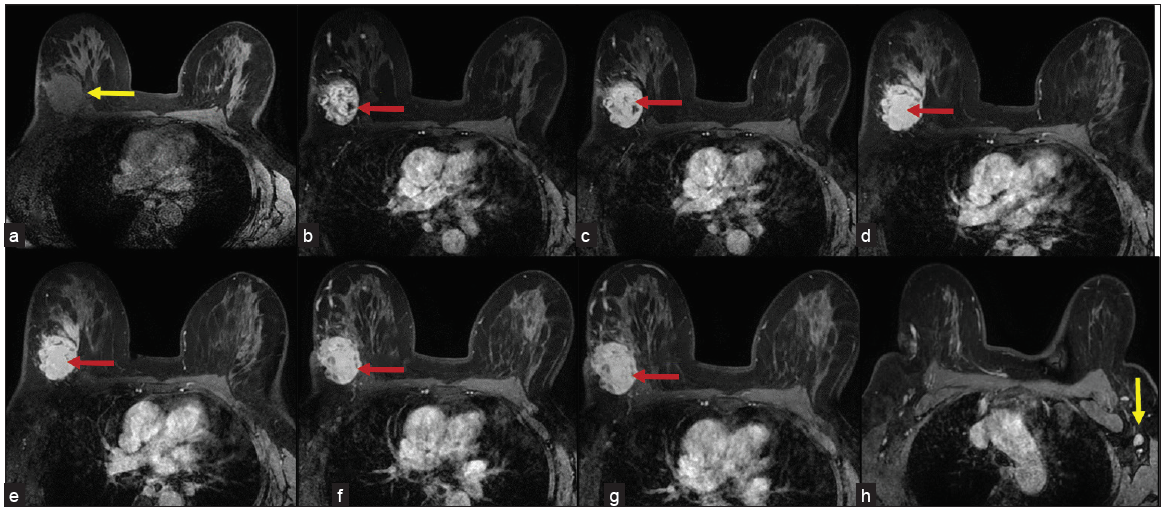
- (a) Precontrast fat suppressed T1 images showing a hypointense lesion (yellow arrow). (b-g) Images of postcontrast fat suppressed T1 images from phase 1 to phase 6 showing intensely enhancing lesion (red arrows) with few non enhancing areas. (h) Enhancing suspicious level I axillary lymph node (yellow arrow) with nodular cortical thickening in the left axilla.
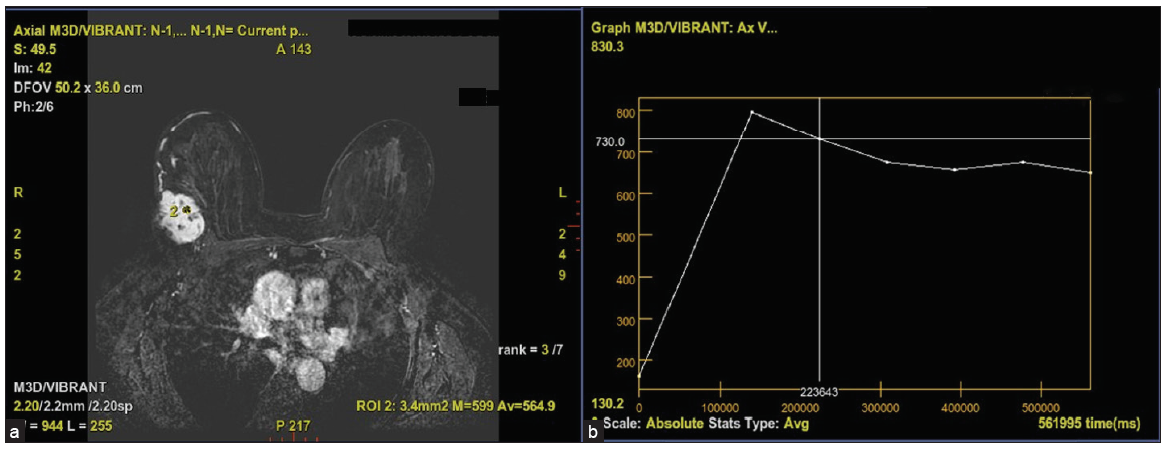
- (a) Figure showing contrast enhanced post processed image with selected ROI. (b) Figure showing type III kinetic curve demonstrating initial rapid uptake and washout in delayed phase. ROI: region of interest.
The patient underwent USG guided biopsy using a 16G needle and 4 sample bits were taken. There was a significant bleed from the site of the biopsy for which gel foam was injected along the tract. Histopathological analysis revealed an Intermediate grade of Angiosarcoma of the breast. The lesion is strongly positive for CD 34 and negative for pan-cytokeratin [Figure 5].
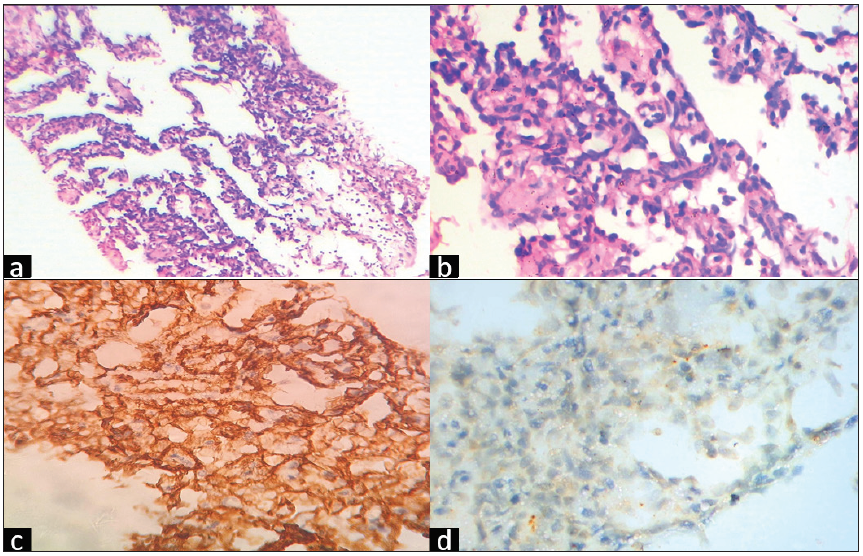
- (a) Low Power H&E (100x) Tumor shows branching dilated vascular channels lined by plump endothelial cells. (b) High power H&E (400x) Tumor cells show hyperchromatic nucleus and focal tufting of endothelial cells. (c) IHC CD34 (400x) - Strong positive in tumor cells. (d) IHC PanCK (400x) - Negative in tumor cells. H&E: hematoxylin and eosin, IHC: immunohistochemistry, CD: cluster of differentiation.
Discussion
Angiosarcoma is one of the rare tumors of the breast, accounting for about 1% of all soft tissue breast tumors.[1] It usually occurs during 3rd and 4th decades of life[2] in contrast to other breast malignancies which occur later in life. In our case, the age of the patient is in the 6th decade.
They can present as primary tumors, or arise secondary to irradiation or lymphedema who underwent treatment for breast cancer.[3] Angiosarcomas present with variable features on ultrasonography and can be seen as well-circumscribed or poorly marginated-masses. They can be hypoechoic or have heterogeneity depending on their vascular channels or cellular components.
Typical of other malignancies, the margins may not often be angular and posterior shadowing is not often found. Hypervascularity is usually seen on Doppler.[3] MRI does better characterization of angiosarcoma. Low-grade sarcomas are generally large and lobulated with borders being indistinct. Lesions are T1 hypointense and T2 hyperintense. In more aggressive malignancies, on contrast administration, there is rapid enhancement with washout pattern which shows a type 3 curve[3-5] and large draining vessels can also be seen.[6] The diagnosis prior to surgery, either by FNA (Fine needle aspiration) or NCB, is often difficult.[7]
Primary angiosarcoma has three grades. Low grade has multiple anastomosing vessels invading the adjacent breast tissue. The second one is of intermediate grade which shows solid neoplastic vascular growth with the mitotic rate being high. Third is high-grade malignancy, which contains frank areas of sarcomatous tissue with necrotic, hemorrhagic, and infarcted areas.
Invasive ductal or lobular carcinomas can be differentiated from angiosarcomas using IHC biomarkers. The presence of CD 31 and CD 34 with absent cytokeratin usually confirms the diagnosis.[4,8] The most specific IHC biomarker for endothelial differentiation is CD 31 and most sensitive biomarker is CD 34.
Angiosarcoma carries worse prognosis as it has tendency to progress and metastasize.[9] Mastectomy is the surgical method of choice for the treatment of this tumor.[10]
In our case, the imaging features are highly nonspecific with the lesion being heterogeneously hypoechoic with lobulated margins, showing posterior acoustic enhancement and significant internal vascularity. On MRI, it is hypointense on T1, hyperintense on T2, STIR, has multiple flow voids, showing diffusion restriction and intense contrast uptake with type III curve in a few areas and type II in the other. Angiosarcoma is best characterized by using contrast-enhanced MRI and the final diagnosis can only be established by using histopathology because of the nonspecific imaging characteristics of angiosarcoma.
Conclusion
Primary angiosarcoma of the breast can often be missed radiologically and pathologically because of its non-specific imaging features. MRI can assess the malignant nature of the lesion compared to the conventional modalities and therefore, MRI must be given high priority to assess the lesion which can be further confirmed by using histopathology. As angiosarcoma is one of the rarest and most aggressive malignancies, the diagnosis should not be delayed and regular follow-up of patients is required to rule out metastasis.
Ethical approval
The research/study approved by the Institutional Review Board at NRI Academy of Sciences, number IEC NRIMC 055/2022, dated 23 Sep 2022.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
References
- Angiosarcoma of the breast: A difficult surgical challenge. Breast. 2012;21:584-9.
- [CrossRef] [PubMed] [Google Scholar]
- Mammary angiosarcomas: Imaging findings in 24 patients. Radiology. 2007;242:725-34.
- [CrossRef] [PubMed] [Google Scholar]
- Primary angiosarcoma of the breast: A case report. Diagn Pathol. 2013;8:66.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Breast angiosarcoma secondary to phyllodestumour. BMJ Case Rep. 2012;2012:bcr2012007545.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Vascular tumors of the breast: Mammographic, sonographic, and MRI appearances. AJR Am J Roentgenol. 2005;184:331-8.
- [CrossRef] [PubMed] [Google Scholar]
- A case of angiosarcoma of the breast. Jpn J Clin Oncol. 1997;27:91-4.
- [CrossRef] [PubMed] [Google Scholar]
- Angiosarcoma after breast-conserving therapy. Cancer. 2003;97:1832-40.
- [CrossRef] [PubMed] [Google Scholar]
- Clinicopathological features of breast angiosarcoma: A 16-years single-institution experience. Int J Surg Case Rep. 2017;37:211-5.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Primary breast angiosarcoma: A rare presentation of rare tumor - case report. BMC ClinPathol. 2017;17:17.
- [Google Scholar]







